Photolon®
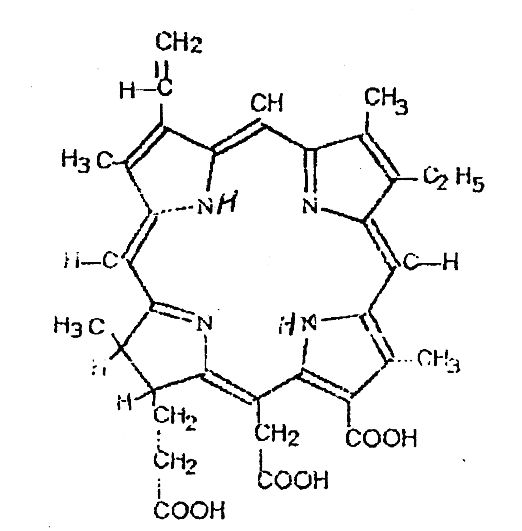
Photolon® is a agent for PDT ( 1, 3, 5, 8 - tetramethyl-4-ethyl-2-vinyl-klorin-6-carbonic-acetic-7-propion acid sodium-vapor salt) 0.1 for intravenous injections designed and produced on pharmaceutical company "Belmedpreparaty" Belarus.
CATEGORY
Antineoplastics.
Photosensitizer for photodynamic therapy and diagnosis
DESCRIPTION
Photolon comprises a molecular complex of a sodium salt of chlorin e6 and its derivatives with low-molecular polymer component of intravenous drugs.
Photolon is a dry scentless porous substance of a dark-green color, soluble in water and salt solutions (25 mg, 50 mg, 100 mg vials)
CLINICAL PHARMACOLOGY
Mechanism of action
The essential feature of photodynamic therapy is the presence of a photosensitizer, which upon its administration into the organism selectively accumulates in a certain type of tissue, and upon irradiation with light of a certain wavelength, produces particles toxic to this type of tissue.
The initial stage includes photoactivation of the photosensitizer by its irradiation by light with a certain wavelength. This is followed by energy transfer from the excited photosensitizer molecule to molecular oxygen present in tissues. This process leads to production of singlet oxygen which interacts with lipids and other components of cell membranes thus leading to disruption of their integrity and, as a result, to the cell death. The second possible mechanism of the photosensitizer effect can be connected with production of free radical particles. These particles can be created as a result of an interaction of an excited photosensitizer molecule immediately with molecular components of the cell, of with an oxygen molecule. The laser treatment induces a photochemical, not a thermal, effect.
Photolon is a second-generation photosensitizer showing a high therapeutic activity.
Photolon is characterized by strong absorption band in the long-wavelength region, which allows using light from the long-wavelength part of the visible range which penetrates deeper into biological tissues, and thus provide a possibility of treatment of relatively massive and deep-lying tumors. Upon intravenous administration, Photolon is highly tumorotropic, possess a high uptake rate in target tissues, have a low phototoxicity, and is virtually completely removed from the body during several days.
PRECLINICAL STUDIES
Effect of Photolon on the depth and frequency of the complete tumor necrosis
a) (sarcoma M-1) in the rat
Photolon at the dose of 2.5 mg/kg was accompanied by light irradiation of 100 J/cm2; the depth of tumor necrosis comprises 11.6±0.5 mm. The frequencies of the complete tumor necrosis is 100%.
b) alveolar liver cancer RS-1in the rat
Photolon at the dose of 2.5 mg/kg was accompanied by light irradiation of 50 J/cm2, the depth of tumor necrosis comprises 12.2±0.7 mm. The complete tumor necrosis is achieved in 100% of cases.
CLINICAL STUDIES
Controlled randomized multicentral phase I and phase II trials of Photolon were carried out at the Medical Radiological Scientific Center (Obninsk, Russia) and Scientific Research Institute for Oncology and Medical Radiology (Minsk, Belarus). PDT with Photolon (1.5–3.0 mg/kg) was carried out for 82 patients (61 – malignant skin neoplasm, 10 – malignant lip neoplasm, 5 – breast cancer with skin metastases, and 6 – malignant melanoma with skin metastases). The complete and partial destruction of malignant tissue was obtained in 85–95% and 5–15% of cases, respectively. Additionally, Photolon was applied in treatment of Kaposi’s sarcoma.
The accumulation level of Photolon in the tumour tissue is 2–20 times higher compared to normal tissue. The above quantity depends on a number of factors including, but not limited to, the photosensitizer dose, and tumour growth stage and rate. The accumulation level of Photolon in tumour tissue increases with the photosensitizer dose administered. A higher accumulation level of Photolon in tumour was found to be characteristic of fast-growing tumours. One of the important advantages of Photolon over other photosensitizers is its fast elimination from the organism: only trace amounts of the preparation are found 24 h after its intravenous injection.
INDICATIONS TO ADMINISTRATION
Photolon is applied for photodynamic therapy of basal-cell skin cancer, flat-cell skin cancer, metatypical skin cancer, flat-cell cancer of the lip, intra- and subcutaneous metastases of breast cancer, intracutaneous metastases of melanoma, and Caposi’s sarcoma; photodynamic therapy of mucous membrane cancer (vulva cancer, uterus cervix cancer, and rectum cancer).
Photosensitizer is applied for photodynamic diagnosis of neoplasms.
CONTRAINDICATIONS
Hypersensitivity to the product, cardiovascular diseases (decompensated), liver and/or renal insufficiency. Pregnancy. Nursing mothers.
Not administered in pediatric practice.
DOSAGE
Photolon should be administered as a slow intravenous injection (dropwise) over 30 minutes at 2.5-3 mg/kg body weight. Reconstitute the adequate dose of Photolon in 200 ml 0.9% Sodium Chloride Injection, shake well until dissolved. Do not mix Photolon with other drugs in the same solution.
Initiate 660-670 wavelength laser light delivery to the patient 3-4 hours following injection of Photolon. A total light dose of 100 - 600 J/cm2 during one or two sessions of laser treatment of one or several areas is administered, depending on the clinical form and morphology of the malignant neoplasm, and on the infiltration depth.
INTERACTIONS
Concominant use of local anesthetics is not recommended.
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs (including analgesic drugs) concomitantly with Photolon.
Antioxidants, Bien, Reparef-2 Ointment could facilitate early epithelization of tissue damage, enhance connective tissue growth, reduce pain.
ADVERSE EFFECTS
Most toxicities are associated with local effects in the region of illumination. Fever and pain in the region of lazer illumination may occur. In patients with cardiovascular diseases sometimes reported hypertension.
Systemically induced effects associated with photodynamic therapy with Photolon is mild photosensitivity. Photosensitivity reactions as a rule are associated with disturbance of precausions to avoid sunlight and bright indoor light (mild hyperemia, edema without pigmentation).
POSSIBLE COMPLICATIONS AND PRECAUTIONS
General
Photosensitivity
All patients who receive Photolon must observe precautions to avoid exposure of skin and eyes to direct sunlight or bright indoor light. Photolon should be administered in darkened room.
Pregnancy
Photolon should not be used during pregnancy.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because potential for serious adverse reactions in nursing infants from Photolon, women receiving Photolon must not breast feed.
Pediatric Use
Safety and effectiveness in children have not been established.
Use in elderly patient
There was no apparent difference in effectiveness or safety in patients over 60 years of age compared to younger people. Dose modification based upon age is not required.
Sunscreen creams should be administered following administration of Photolon.
OVERDOSAGE
There is no information on overdosage situation involving Photolon.
LD50 of Photolon is not less than 140 mg/kg in the rat.
In animals toxic effects include dyspnoea, tachycardia, hypertension, excitation, following central nervous system depression, hypotermia.
DOSAGE FORMS
Photolon for ingection is supplied as a freeze-dried cake or powder as 25 mg, 50 mg, and 100 mg vials.
STORAGE
Store at temperature not more + 10°C, in dark (protected from light) place.